 
A similar effect is seen when the anion becomes larger in a series of compounds with the same cation.Īrrange GaP, BaS, CaO, and RbCl in order of increasing lattice energy. Na+(g) + Cl(g) NaCl(s) During the formation of NaCl, both the cation and anion are in a gaseous state. Because r 0 in Equation 4.2.1 is the sum of the ionic radii of the cation and the anion ( r 0 = r + + r −), r 0 increases as the cation becomes larger in the series, so the magnitude of U decreases. Lattices sodium chloride We have already mentioned that for sodium chloride approximately 1.78 times as much energy is released when the crystal lattice forms as when ion pairs form. Lattice energy can also be defined as the amount of energy released when gaseous cation and anion combine and form one mole of an ionic compound. This effect is illustrated in Figure 4.2.2, which shows that lattice energy decreases for the series LiX, NaX, and KX as the radius of X − increases. For example, the calculated value of U for NaF is 910 kJ/mol, whereas U for MgO (containing Mg 2 + and O 2− ions) is 3795 kJ/mol.īecause lattice energy is inversely related to the internuclear distance, it is also inversely proportional to the size of the ions. For example, if we consider NaCl (6:6) ionic crystal then c6, and if CsCl (8:8) ionic. 
You should talk about lattice formation enthalpy if you want to talk about the amount of energy. c is the first order coordination number concerning our reference ion. Source: Data from CRC Handbook of Chemistry and Physics (2004).īecause the lattice energy depends on the product of the charges of the ions, a salt having a metal cation with a +2 charge (M 2 +) and a nonmetal anion with a −2 charge (X 2−) will have a lattice energy four times greater than one with M + and X −, assuming the ions are of comparable size (and have similar internuclear distances). For NaCl, the lattice dissociation enthalpy is +787 kJ mol-1. Lattice H denotes the molar lattice enthalpy. Where: LatticeU denotes the molar lattice energy. Energies of this magnitude can be decisive in determining the chemistry of the elements. The molar lattice energy of an ionic crystal can be expressed in terms of molar lattice enthalpy, pressure, and change in volume via the following equation: LatticeU LatticeH pVm. Hence the value of lattice energy is always positive and endothermic. Representative values for calculated lattice energies, which range from about 600 to 10,000 kJ/mol, are listed in Table 4.2.1. Hint: Lattice energy is the energy required to break apart an ionic solid and convert its component atoms into gaseous ions. The value of the constant k′ depends on the specific arrangement of ions in the solid lattice and their valence electron configurations, topics that will be discussed in more detail in the second semester. We see from Equation 4.4 that lattice energy is directly related to the product of the ion charges and inversely related to the internuclear distance. As before, Q 1 and Q 2 are the charges on the ions and r 0 is the internuclear distance. U, which is always a positive number, represents the amount of energy required to dissociate 1 mol of an ionic solid into the gaseous ions. The equation is: Lattice enthalpy Heat of formation- Heat of sublimation- (1/2 Dissociation energy) - (sum of Ionization energies)- (sum of Electron Affinities) How do you calculate. Therefore lattice energy for NaCl is 756 KJ per mol.\) They do not easily deform, as well as they melt at relatively high temperatures. The solvation energy is sufficient to overcome the attraction between the ions in the crystal lattice. Ionic compounds are usually rigid, brittle, crystalline substances with flat surfaces in a lattice form. It is, electrons transfer from the metal to the non-metal. Recall that the reaction of a metal with a non-metal usually produces the ionic compound. Now we calculate the lattice energy of sodium chloride, In Single Step: Na (s) + 1/2 Cl 2(g) NaCl (s) H f. 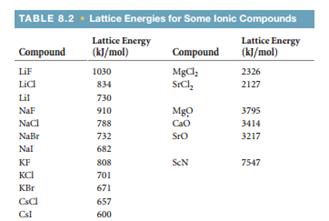
The principle of Born Haber cycle is Hess’s law (energy involve in single step energy involved in different steps) H f H sub + I.E + H diss + E.A + U. The lattice energy for NaCl is: NaCl (s) Na + (g) + Cl (g) H lattice +787 kJ/mol The process absorbs energy, and is highly endothermic. The energy released in several steps is called Lattice energy. Lattice energy can be a very complicated process but it can be easily understood by using Coulomb’s law. Lattice energy, E lattice is the energy required to separate one mole of a solid ionic compound into its gaseous ions. The attraction of the two ions releases energy, so this process is of an exothermic kind. Na + (g) + Cl-(g) NaCl(s) H o -787.3 kJ: If we consider just the first four steps in this reaction, Hesss law suggests that it 5.16 takes 376.0 kJ/mol to form Na + and Cl-ions from sodium metal and chlorine gas. Lattice energy refers to the energy which is released while two oppositely charged gaseous ions attract to each other and form an ionic solid. This is a strongly exothermic reaction, for which H o is equal to the lattice energy of NaCl. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
